Oseltamivir

Administered as ethyl ester (the prodrug), since it is orally absorbed better than the free acid, which is the active drug.
Tamiflu®
As an analog of sialic acid, it binds the active site in viral neuraminidase.
Laninamivir

The octanoate is a very lipophylic compound used as long-term antiviral –a `depot´ formulation- and slowly releases the alcohol which is the active drug. It was marketed in 2010. It is administered intranasally in a single dose.
Inavir®
Zanamivir
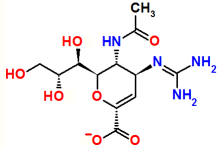
Relenza®
As an analog of sialic acid, it binds the active site in viral neuraminidase.
Peramivir
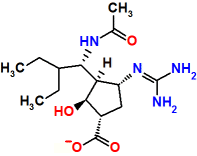
It was marketed in 2010.
PeramiFlu®
It has 5 chiral centres:
(1S,2S,3S,4R)-